 
Some stable helium-3 is produced in fusion reactions from hydrogen, but it is a very small fraction, compared with the highly energetically favorable production of helium-4. In a similar way, the particular energetic stability of the helium-4 nucleus, produced by similar effects, accounts for the ease of helium-4 production in atomic reactions involving both heavy-particle emission and fusion. This arrangement is thus energetically extremely stable for all these particles, and this stability accounts for many crucial facts regarding helium in nature.įor example, the stability and low energy of the electron cloud of helium causes helium's chemical inertness (the most extreme of all the elements), and also the lack of interaction of helium atoms with each other (producing the lowest melting and boiling points of all the elements). Adding another of any of these particles would require angular momentum, and would release substantially less energy (in fact, no nucleus with five nucleons is stable). This symmetry reflects similar underlying physics: the pair of neutrons and the pair of protons in helium's nucleus obey the same quantum mechanical rules as do helium's pair of electrons (although the nuclear particles are subject to a different nuclear binding potential), so that all these fermions fully occupy 1s orbitals in pairs, none of them possessing orbital angular momentum, and each canceling the other's intrinsic spin. High-energy electron-scattering experiments show its charge to decrease exponentially from a maximum at a central point, exactly as does the charge density of helium's own electron cloud. The nucleus of the helium-4 atom is identical to an alpha particle. Stability of the 4He nucleus and electron shell In an experiment involving the use of exotic helium atoms where an atomic electron was replaced by a muon, the nucleus size has been estimated to be 1.67824(83) fm. The size of the 4He nucleus has long been known to be in the order of magnitude of 1 fm. However, numerical approximations of the equations of quantum mechanics have given a good estimate of the key atomic properties of helium-4, such as its size and ionization energy. The helium atom is the second simplest atom (hydrogen is the simplest), but the extra electron introduces a third "body", so the solution to its wave equation becomes a " three-body problem", which has no analytic solution. Helium-4 also exists on the Moon and - as on Earth - it is the most abundant helium isotope. It is theorized that at 0.2 K and 50 atm, solid helium-4 may be a superglass (an amorphous solid exhibiting superfluidity). The superfluid behavior is now understood to be a manifestation of Bose–Einstein condensation, which occurs only with collections of bosons. The total spin of the helium-4 nucleus is an integer (zero), and therefore it is a boson (as are neutral atoms of helium-4). This strange behavior is a result of the Clausius–Clapeyron relation and cannot be explained by the current model of classical mechanics, nor by nuclear or electrical models - it can only be understood as a quantum-mechanical phenomenon. In this state and situation, it is called a " Rollin film". For example, if superfluid helium-4 is kept in an open vessel, a thin film will climb up the sides of the vessel and overflow. When liquid helium-4 is cooled to below 2.17 K (−270.98 ☌), it becomes a superfluid, with properties that are very unlike those of an ordinary liquid. Helium-4 makes up about one quarter of the ordinary matter in the universe by mass, with almost all of the rest being hydrogen. However, primordial helium-4 is largely absent from the Earth, having escaped during the high-temperature phase of Earth's formation. While it is also produced by nuclear fusion in stars, most helium-4 in the Sun and in the universe is thought to have been produced by the Big Bang, and is referred to as " primordial helium". Its nucleus is identical to an alpha particle, and consists of two protons and two neutrons.Īlpha decay of heavy elements in the Earth's crust is the source of most naturally occurring helium-4 on Earth, produced after the planet cooled and solidified. 
It is by far the more abundant of the two naturally occurring isotopes of helium, making up about 99.99986% of the helium on Earth. 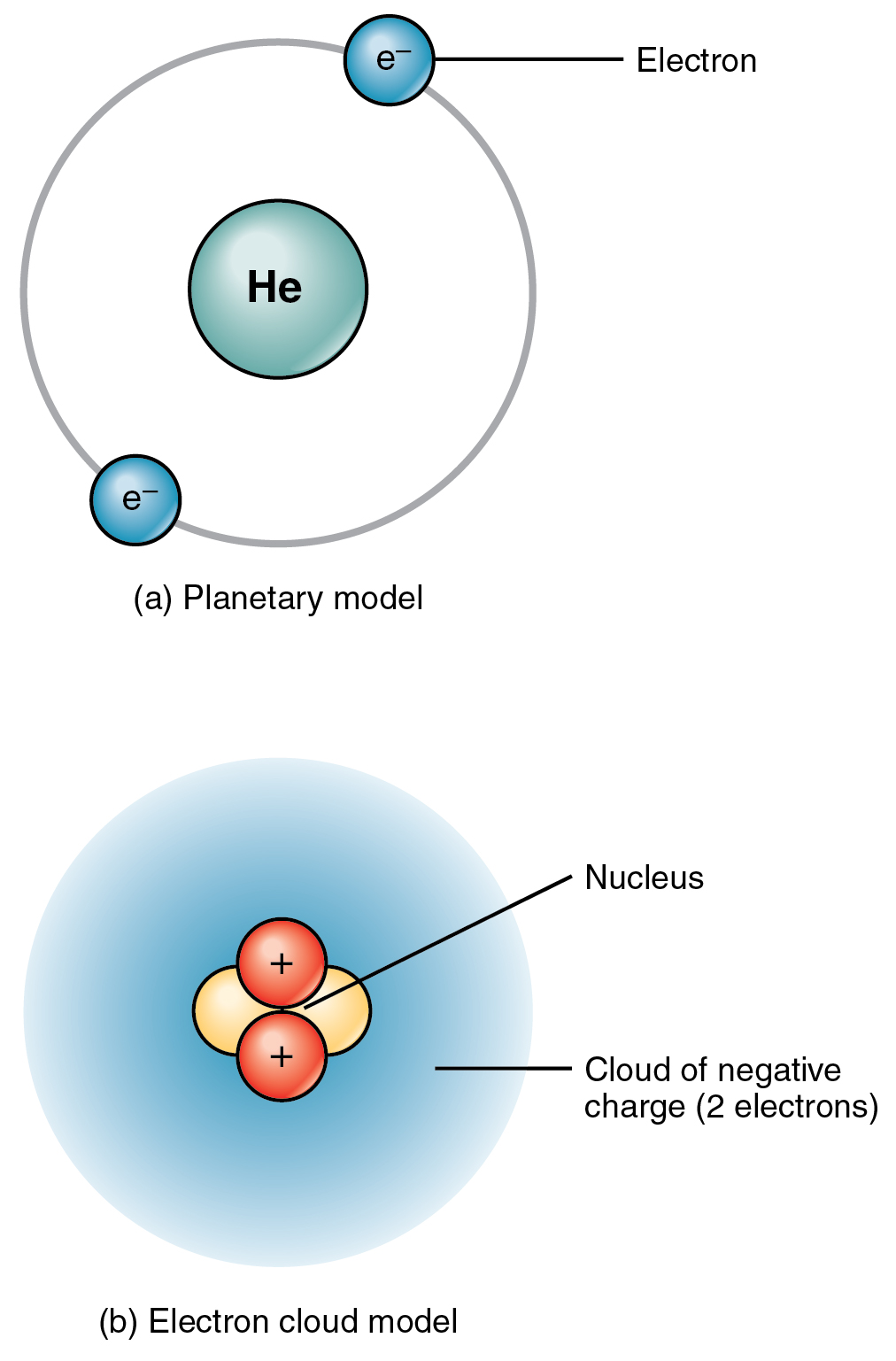
) is a stable isotope of the element helium. The nucleus (upper right) in helium-4 is in reality spherically symmetric and closely resembles the electron cloud, although for more complicated nuclei this is not always the case. Depicted are the nucleus (pink) and the electron cloud distribution (black). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
